High vs. gradually increasing energy nutrition in the early phase of acute pancreatitis (GOULASH trial)
Márta K et al, Gut. 2026;75(5):995-1002
Based on this early terminated trial, a high-energy strategy for early nutrition in pancreatitis does not decrease mortality/severity, but potentially increases organ failure and pain relapse rate.
Background: Acute pancreatitis (AP) is among the most common gastrointestinal diseases requiring hospitalisation, often with severe outcomes and no disease-specific therapy. Nutritional support has been proven to improve outcome, but little is known regarding optimal timing and composition.
Objective: This clinical trial aimed to compare high (30 kcal/kg/day, high energy (HE)) versus gradually increasing energy (0 increased to 30 kcal/kg/day over 4 days, low energy (LE)) strategies for enteral nutritional support in AP.
Design: This was a multicentric, double-blind, randomised clinical trial, enrolling patients with AP regardless of predicted severity (January 2017 to April 2023). The primary outcome was a combination of mortality and severe acute pancreatitis (Revised Atlanta Criteria); secondary outcomes included severity, rate of infection, organ failure and pain relapse. Interim analysis was planned after 50% enrolment. The Benjamini-Hochberg false discovery rate (FDR) method was used to correct p value for multiple testing.
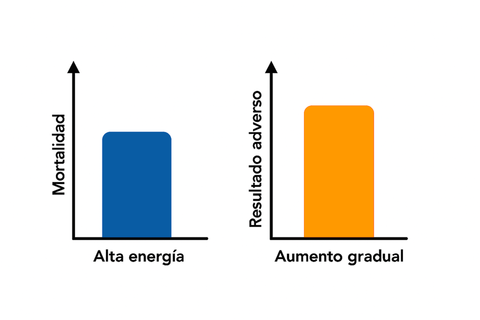
Results: The trial was stopped early after enrolling 636 patients. Interim analysis showed that the primary outcome showed no difference between groups in the modified intention-to-treat (mITT) population (HE: 28/312, 9.0% vs. LE: 18/307, 5.7%, p(uncorrected/corrected) = 0.19/0.42). Secondary outcomes showed no difference in the mITT analysis. Without correction for multiplicity testing, results favoured a low gradual energy strategy in terms of organ failure (HE: 52/312, 16.7% vs. LE: 28/307, 9.1%, p(uncorrected) = 0.007) and pain relapse (80/312, 27.1% vs. 54/307, 19.0% p(uncorrected) = 0.03) but showed no differences between groups after correction for multiple testing (p = 0.13 and p = 0.23, respectively). It was determined that the superiority of the intervention would not be shown even with an increased sample size, and thus the trial was terminated based on a post hoc decision on ethics and futility.
Conclusion: Based on this early terminated trial, a high-energy strategy for early nutrition in pancreatitis does not decrease mortality/severity, but potentially increases organ failure and pain relapse rate.